Ask about FDA oncology approvals, clinical trials, and treatment guidelines.
Latest FDA Oncology Updates
FDA Accepts NDA for TLX101-Px (Pixclara®) - PR Newswire
FDA Accepts NDA for TLX101-Px (Pixclara®). (PRNewsfoto/Telix Pharmaceuticals ... cancer), and has assigned a PDUFA goal date of September 11, 2026 ...
April 30, 2026: Meeting of the Oncologic Drugs Advisory Committee
Event Title. April 30, 2026: Meeting of the Oncologic Drugs Advisory Committee Meeting Announcement. April 30, 2026 · On This Page · Agenda · Meeting Materials.
Replimune Receives Complete Response Letter from the FDA
Replimune Receives Complete Response Letter from the FDA for RP1 Biologics License Application for the Treatment of Advanced Melanoma. 10 avr. 2026 17h02 HE | ...
Savara Announces the U.S. Food & Drug Administration (FDA) Has ...
Such statements include, but are not limited to, statements related to the target PDUFA action date. Savara may not actually achieve any of the matters ...
FDA approves nivolumab with chemotherapy for Hodgkin lymphoma
This review was conducted under Project Orbis, an initiative of the FDA Oncology Center of Excellence. Project Orbis provides a framework for concurrent ...
FDA Schedules Public Meeting on the Commissioner's National ...
... Oncology Center of Excellence, as well as a presiding officer. Requests to speak are due by April 24 at 11:59 p.m. The FDA is also soliciting written ...
FDA Oncology Center of Excellence Presents Conversations on ...
FDA Oncology Center of Excellence Presents Conversations on Cancer: Oncology Nurses - Frontliners in Oncology May 6, 2026.
FDA approves relacorilant with nab-paclitaxel for platinum-resistant epithelial ovarian, fallopian tube, or primary peritoneal cancer
On March 25, 2026, the Food and Drug Administration approved relacorilant (Lifyorli, Corcept Therapeutics Inc.), a glucocorticoid receptor antagonist, in combination with nab-paclitaxel for the treatment of adults with platinum-resistant epithelial ovarian, fallopian tube, or primary peritoneal cancer who have received one to three prior systemic treatment regimens, at least one of which included bevacizumab.
Recent Posts
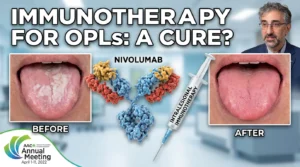
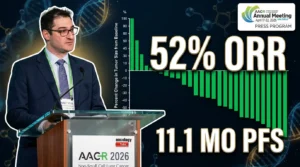
- Could Intralesional Nivolumab Finally End the Era of Disfiguring Oral Surgeries?
- How DNA Methylation Predicts Tissue of Origin in CUP
- CARPRISM Trial: Can Cilta-cel Deliver Universal MRD Negativity and Prevent Progression in High-Risk Smoldering Multiple Myeloma?
- Does QHS-5132 Signal a Biomarker-Agnostic Shift in Ovarian Cancer Treatment?
- Is Alisertib the New Standard for Overcoming Resistance in KRAS G12C-Mutant NSCLC?